The Molecular Interactions of the MAPK Pathway
Hellow guys, Welcome to my website, and you are watching The Molecular Interactions of the MAPK Pathway. and this vIdeo is uploaded by Ribosome Studio at 2018-05-20T18:51:14-07:00. We are pramote this video only for entertainment and educational perpose only. So, I hop you like our website.
Info About This Video
| Name |
The Molecular Interactions of the MAPK Pathway |
| Video Uploader |
Video From Ribosome Studio |
| Upload Date |
This Video Uploaded At 21-05-2018 01:51:14 |
| Video Discription |
An animation about the MAPK pathway and its role in cell division.
Supported by the Zurich University of Applied Sciences/ZHAW Life Science / FM pharmaceutical biotechnology masters program.
Find us on Social Media:
Instagram: https://www.instagram.com/ribosomestudio/ (@ribosomestudio)
Facebook: https://www.facebook.com/RibosomeStudio/ (@ribosomestudio)
LinkedIn: https://www.linkedin.com/company/ribosome-studio/
Google+: https://plus.google.com/u/0/b/116693374515065001139/
https://www.ribosomestudio.com/
Script:
Nevena Hristozova
Twitter: @NevenaHr
http://www.incubatorium.eu/
......................................................................................................
Transcript:
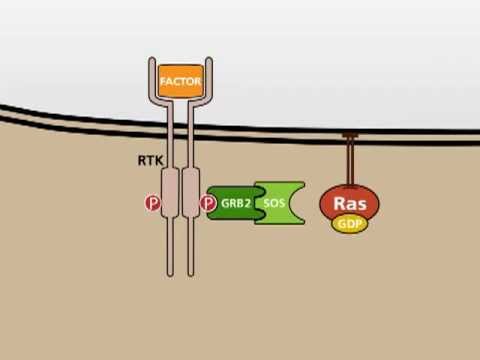
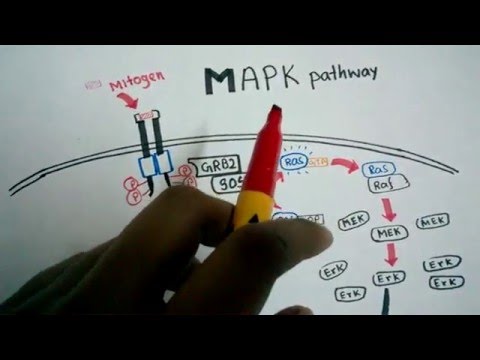
EGF stimulates cell growth and differentiation by binding to EGFR, a transmembrane protein that belongs to the family of receptor tyrosine kinases.
When EGF binds to EGFR, it locks the receptor in a specific conformation allowing a second receptor tyrosine kinase to bind effectively.
In this animation, a HER2 molecule binds EGFR. In contrast to EGFR, HER2 does not need an extracellular ligand like EGF to be able to dimerize.
This allows transphosphorylation to take place on the intracellular domains of EGFR and HER2 thereby activating them.
Only when active, the intracellular domains of EGFR can recruit the downstream signaling molecules of the MAPK cascade - GRB2 and SOS. GRB2 is effectively the mediator protein for the signal from the receptor to the soluble intracellular proteins.
SOS serves as a docking site for RAS. Upon binding of RAS, it is able to exchange the GDP in the cofactor binding pocket with GTP thereby activating RAS.
RAS then dissociates from the activation complex while staying attached to the cell membrane.
In this manner, one GRB2-SOS complex can activate multiple membrane-bound RAS proteins, thereby leading to a signal amplification.
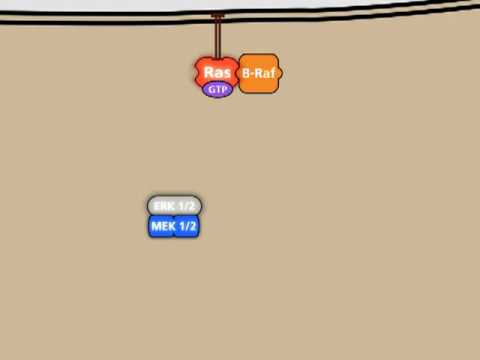
The signal cascade continues with RAF.
In the inactive state two 14-3-3 proteins are bound to RAF, one of them blocking the interaction surface with RAS.
To activate RAF it first needs to get rid of the n-terminally bound 14-3-3 protein.
Separated by 14-3-3 it is possible for RAF to change its shape and bind to RAS.
Multiple activated RAS proteins combined with RAF form a structure called a nanocluster, which can then recruit downstream signaling proteins.
The sole removal of the inhibitor protein 14-3-3 is not enough to activate RAF. Another protein called SRC is needed to phosphorylate RAF to fully activate it.
A scaffolding protein binds RAF and SRC and forms a complex. The scaffold increases the local concentration of RAF and SRC, allowing them to interact and ultimately facilitating the activation of RAF by phosphorylation.
Activated RAF is able to activate the downstream signaling molecules MEK and ERK. Once again, scaffold proteins are required to do that efficiently.
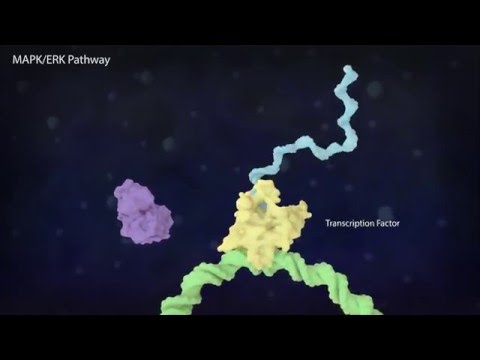
Activated ERK translocates to the nucleus.
In the nucleus, ERK activates MYC - a transcription factor involved in the regulation of 15% of all genes.
Normally MYC is degraded rather quickly in the nucleus by proteasomes. But by activating MYC, the half-life is increased and it can accumulate in the nucleus.
MYC can then form a dimer with MAX. This dimer can subsequently bind specific motifs on the DNA.
Aside from the DNA, MYC can bind histone acetyltransferases. Acetylating the histones makes them negatively charged. Since DNA itself is negatively charged, this causes dissociation of the histones and allows the transcription machinery to access the DNA and start the expression of genes located there.
Another way of influencing the gene expression is by crosslinking two DNA strand via MYC-MAX and MAD-MAX dimers.
With these complex interactions, the docking of a simple extracellular growth factor can cause a shift of the protein expression pattern of an entire cell.
#science #medical #biology |
| Category |
Science & Technology |
| Tags |
MAPK | Science | Medical animation | EGFR | EGF | MAPKK | MAPKKK | SOS | GRB2 | RAF | MEK | ERK | MYC | MAX | MAD-MAX | scientific | Molecules | Protein | MAPK Pathway | MAPK Pathway Animation | mapk signaling pathway | mapk pathway cancer |
More Videos